
While the HHS public health emergency is ending, COVID-19 remains a high priority and the U.S. Food and Drug Administration will remain engaged with numerous activities to protect and promote public health.

Federal, Provincial, Territorial Public Health Response Plan for Ongoing Management of COVID-19
FDA Roundup: May 12, 2023 - US FDA

Science and Engineering Laboratories: Updates from FDA/CDRH - US FDA
Fresh Arrivals: Latest Products in Our Outlet with Great Discounts - Nae Vegan Shoes

AnaBot: Enhance ChatGPT Conversations & Insights

Frontiers China's Public Health Policies in Response to COVID-19: From an “Authoritarian” Perspective

How Penn Medicine Epidemiologists Prepared for Future Pandemics
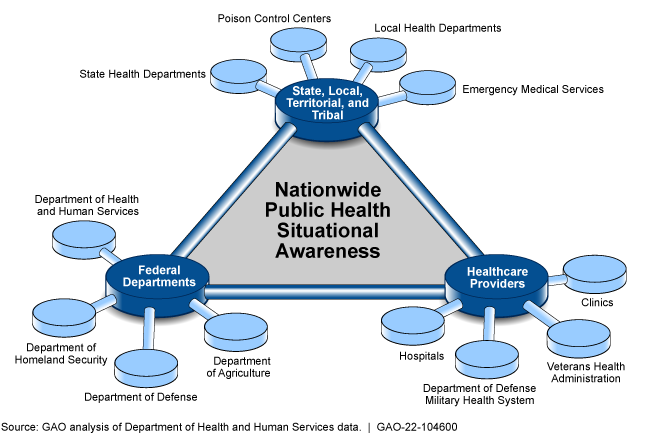
COVID-19: Pandemic Lessons Highlight Need for Public Health Situational Awareness Network
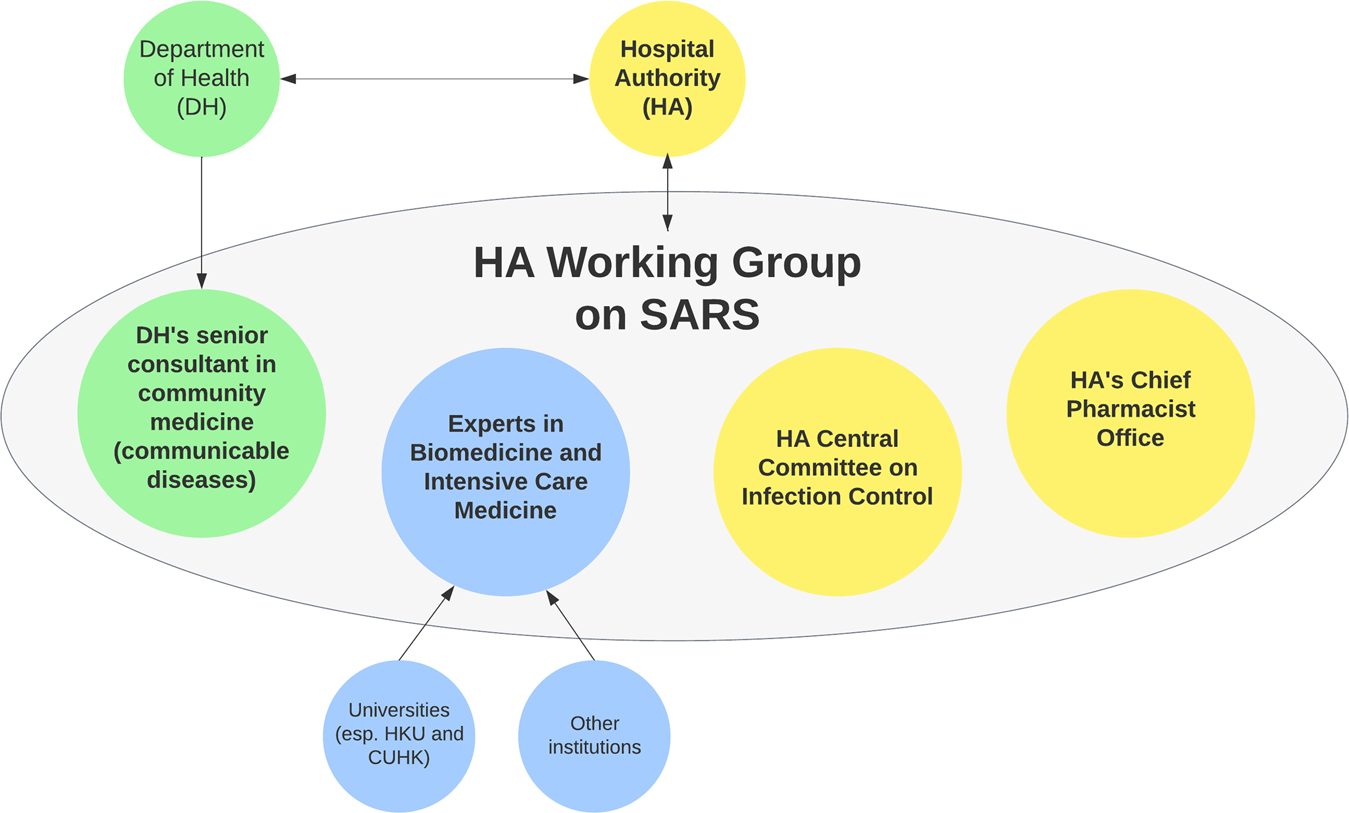
From SARS to COVID-19: the role of experience and experts in Hong Kong's initial policy response to an emerging pandemic

COVID-19 and Human Development: Assessing the Crisis, Envisioning the Recovery
FDA Roundup: May 12, 2023 - US FDA

Public Health Preparedness Capabilities: National Standards for State and Local Planning, State and Local Readiness

First lessons from government evaluations of COVID-19 responses: A synthesis
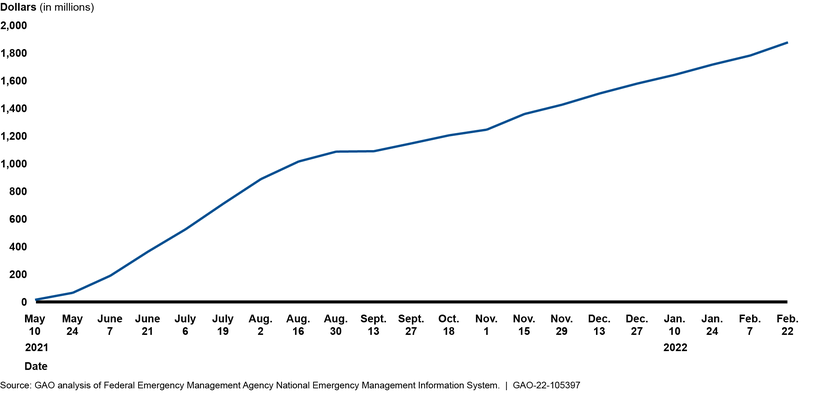
COVID-19: Current and Future Federal Preparedness Requires Fixes to Improve Health Data and Address Improper Payments
Trend Alert: 🪩 Retro Inspired Photos - Archipelago Presets