
Definition: The Universal or Ideal Gas Law describes the relationship between all four properties (pressure, volume, number of moles, and temperature) as well as a gas constant called “R.” NOTE: The Ideal Gas Constant “R” has constant a value of 0.0821 L.atm/mol.K Relation: The relation between pressure (P) volume (V), number of moles (n) and…

Combined and Ideal Gas Law Worksheet by Ms Stricklin Chemistry Corner

Polarity: Dipoles and Dipole Moments

Equation Of State (Ideal Gas), Glenn Research Center
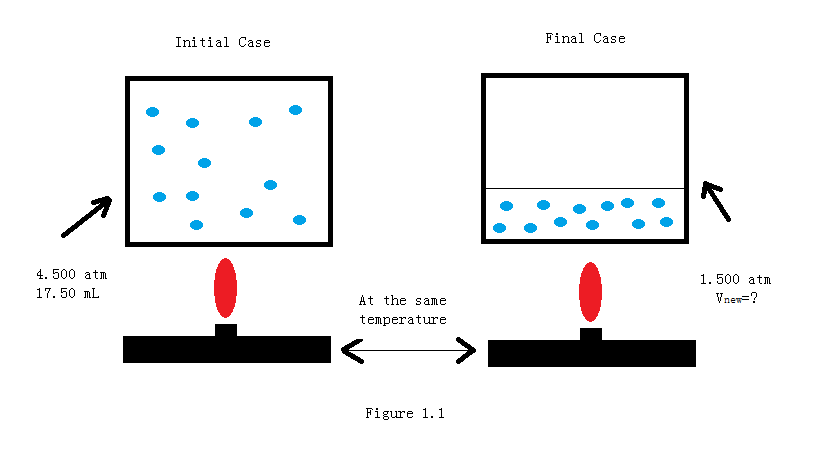
Gas Laws - Overview - Chemistry LibreTexts

6.3: Combining the Gas Laws: The Ideal Gas Equation and the General Gas Equation - Chemistry LibreTexts

Ideal–Universal Gas Law

Standard Temperature and Pressure

Gas Laws Flashcards

Ideal Gas Law Equation Compressibility Of Natural Gas - Chemistry

SOLUTION: Ideal Gas law and Universal Gas Constant Questions and Answers - Studypool
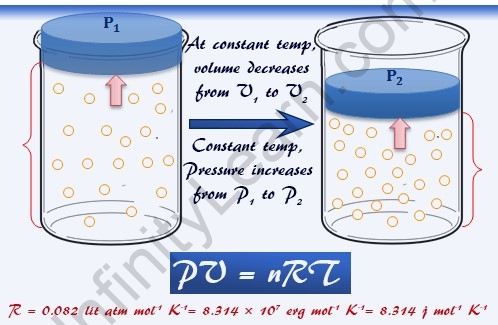
Dimensions Of Universal Gas Constant - Infinity Learn by Sri Chaitanya
Gas Laws Revision Table Worksheet + Answers by ACE Science Resources

Ideal Gas Law: Calculation of Universal Gas Constant, General Chemistry

ANESTHESIA EQUIPMENT AND GAS LAW REVIEW - ppt download