By A Mystery Man Writer

Accelerated aging is a medical package testing method that can accurately assess packaging durability by simulating environmental factors such as heat and humidity.
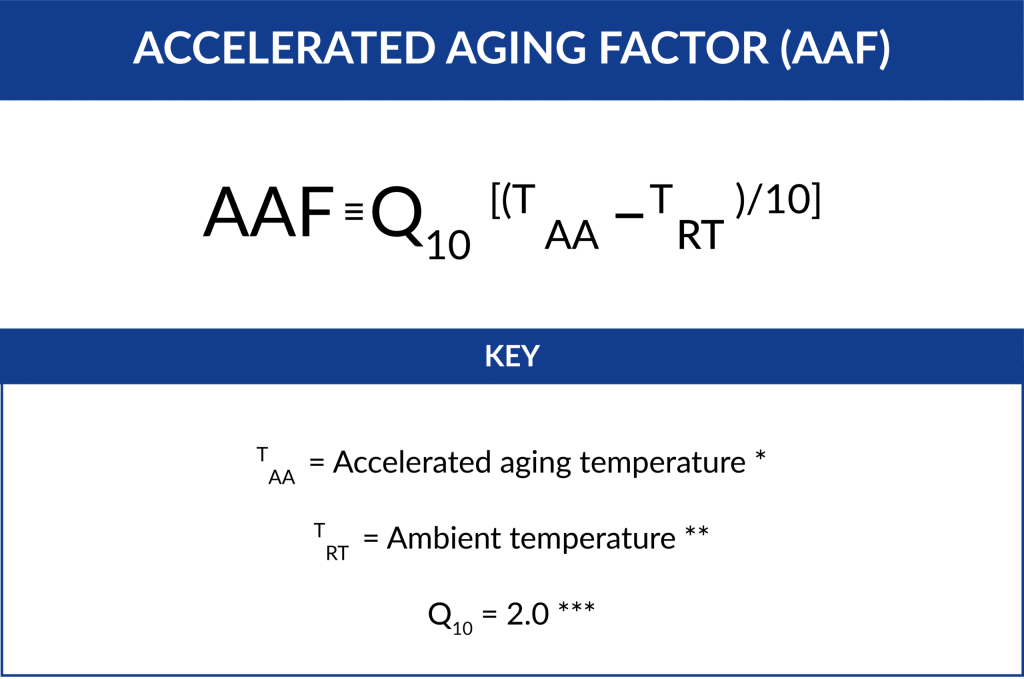
Packaging Validation Process and Guidelines: Accelerated Aging
Standard Practice for Determination of Expiration Dating for Medical Gloves

ASTM D7160-16(2023) - Standard Practice for Determination of Expiration Dating for Medical Gloves

Learning Center - Life Science Outsourcing, Inc.
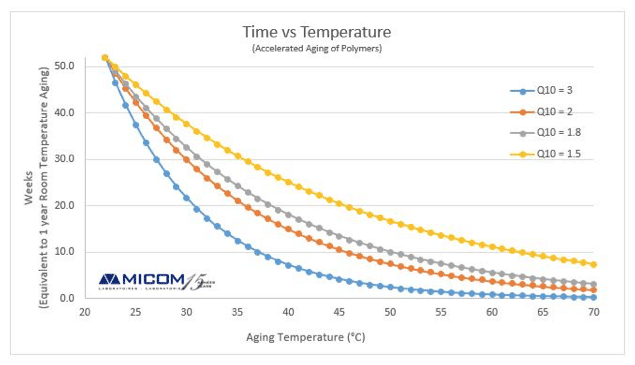
ASTM F1980 Is An Accelerated Aging Test Used For Medical Devices

Overview of Accelerated and Real Time Aging's Role in Package Validation

Climatic Simulate Solar Panel Weather Resistance UV Accelerate Aging Test Equipment Machine - China Humidity Chamber, Environmental Chamber
The Accelerated Aging Study simulates the effects of aging, weathering and handling by subjecting sterile packaged product to conditions considered

Accelerated Aging Study
The Shelf Life Procedure defines the required testing needed to support medical device shelf life claims. The Shelf Life Procedure provides

Product Stability Shelf-Life Procedure | ISO 13485 | FDA QSR Compliant

MycoScience - Accelerated aging

Accelerated Aging Testing [Medical Device Accelerated Aging]