By A Mystery Man Writer

Answer to The compressibility factor is Z = PV/R_g T. Evaluate

Compressibility factor - Wikipedia

PDF) Predicting the compressibility factor of natural gases containing various amounts of CO2 at high temperatures and pressures

Gas compressibility factor Z: Ideal gas vs Real gas

PDF) Theoretical Assessment of Compressibility Factor of Gases by Using Second Virial Coefficient

PDF) Compressibility factor as evaluation parameter of expansion processes in Organic Rankine Cycles
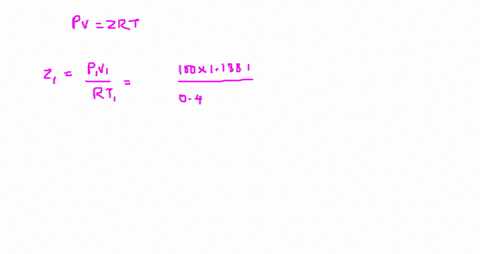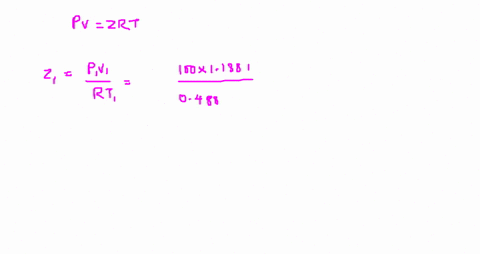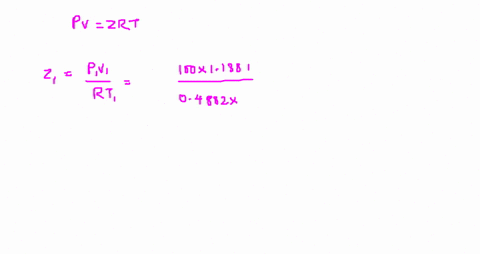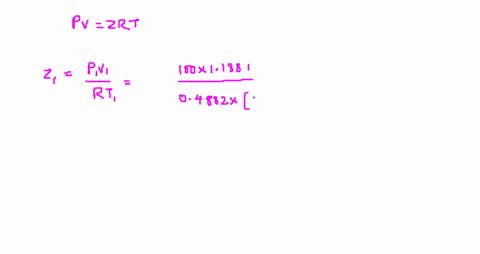
⏩SOLVED:The value of compressibility factor (Z) for this vapour is?…

If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as

The value of compressibility factor (`Z`) for an ideal gas is
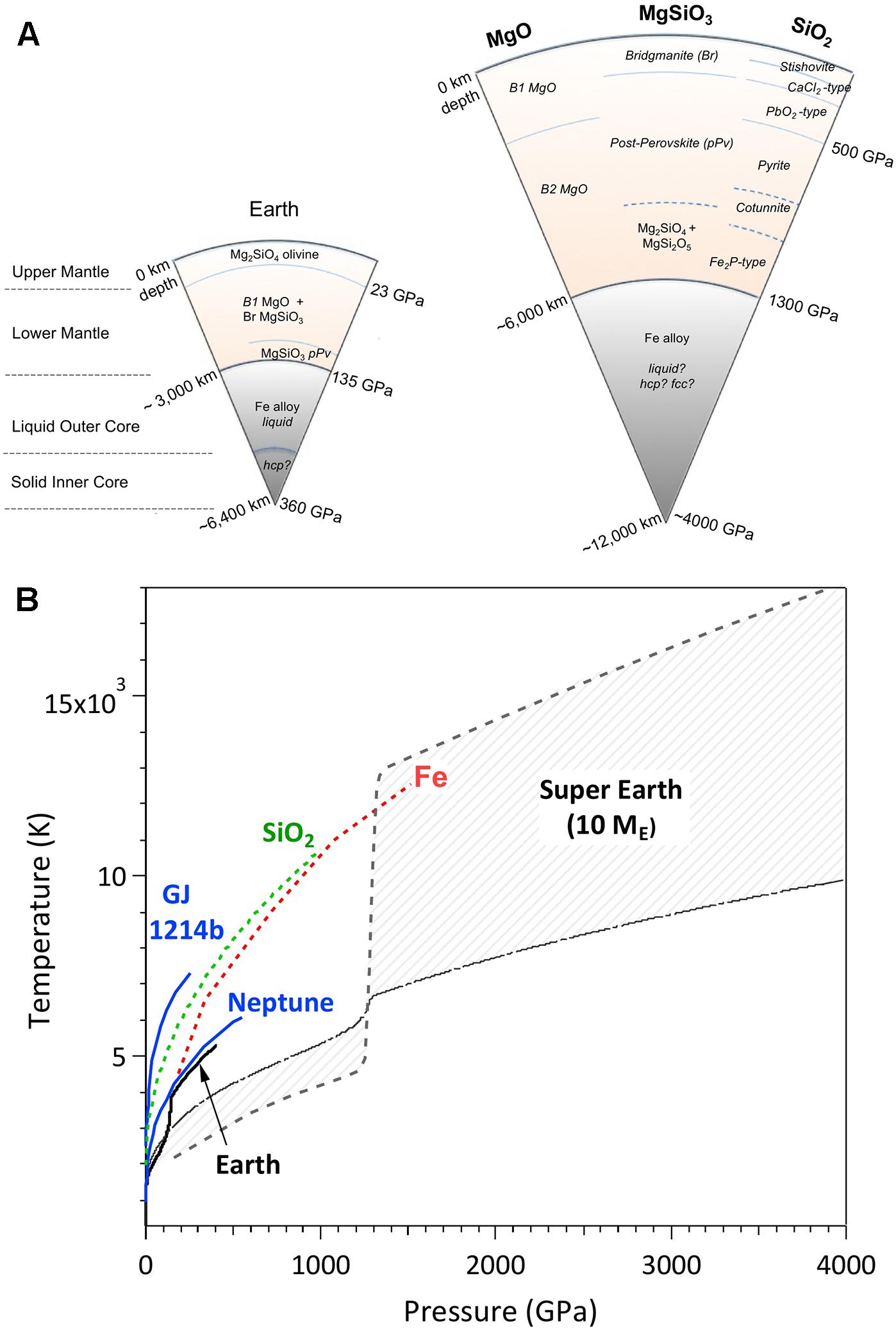
Frontiers Ultra-High Pressure Dynamic Compression of Geological Materials

SOLVED: The compressibility factor, z, is used for predicting the behavior of non-ideal gases. How is the compressibility factor defined relative to an ideal gas? (Subscript c refers to critical value.) a)

Mechanical metamaterials associated with stiffness, rigidity and compressibility: A brief review - ScienceDirect
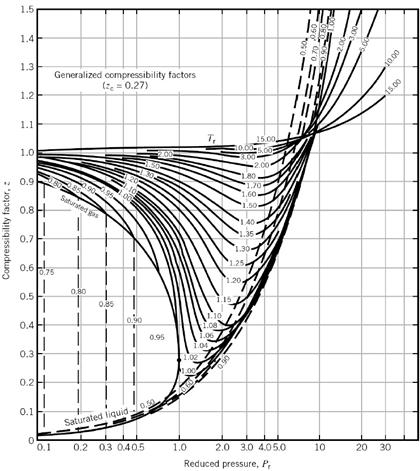
Z= PVm / RT for gases as a function of the reduced

Van der Waals equation - Wikipedia

Compressibility factor z versus 100/V, for several values of Pressure

The value of compressibility factor at the critical state the gas matches with the `Z_(c )` is